Reagent-Controlled Synthesis of the Branched Trisaccharide Fragment of the Antibiotic Saccharomicin B
Published
May 23, 2018
Source
Sameh E. Soliman and Clay S. Bennett
Abstract
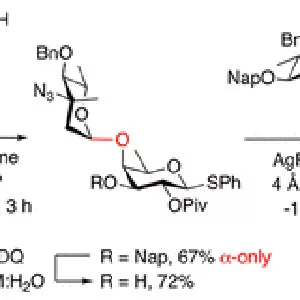
A concise synthesis of a branched trisaccharide, α-l-Dig-(1 → 3)-[α-l-Eva-(1 → 4)]-β-d-Fuc, corresponding to saccharomicin B, has been developed via reagent-controlled α-selective glycosylations. Starting from the d-fucose acceptor, l-epi-vancosamine was selectively installed using 2,3-bis(2,3,4-trimethoxyphenyl)cyclopropene-1-thione/oxalyl bromide mediated dehydrative glycosylation. Following deprotection, l-digitoxose was installed using the AgPF6/TTBP thioether-activation method to produce the trisaccharide as a single α-anomer. This highly functionalized trisaccharide can potentially serve as both a donor and an acceptor for the total synthesis of the antibiotic saccharomicin B.
Posted by
Rima Mycynek

