White Paper: Developing Antimicrobial Drugs for Resistant Pathogens, Narrow-Spectrum Indications, and Unmet Needs.
Published
July 15, 2017
Source
Helen W Boucher, Paul G Ambrose, H F Chambers, Richard H Ebright, Amanda Jezek, Barbara E Murray, Jason G Newland, Belinda Ostrowsky, and John H Rex on behalf of the Infectious Diseases Society of America
The Journal of Infectious Diseases, Volume 216
The Journal of Infectious Diseases, Volume 216, Issue 2, 15 July 2017, Pages 228–236
Abstract
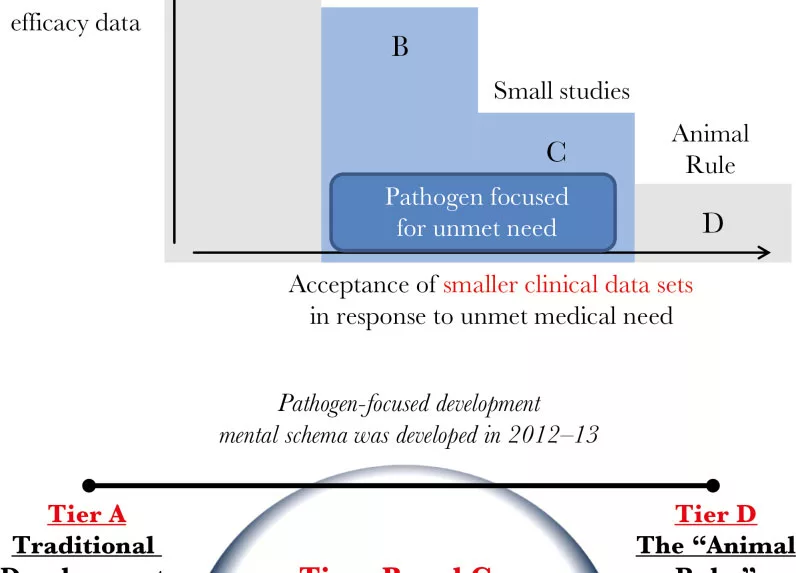
Despite progress in antimicrobial drug development, a critical need persists for new, feasible pathways to develop antibacterial agents to treat people infected with drug-resistant bacteria. Infections due to resistant gram-negative bacilli continue to cause unacceptable morbidity and mortality rates. Antibacterial agents have been historically studied in noninferiority clinical trials that focus on a single site of infection (eg, complicated urinary tract infections, intra-abdominal infections), yet these designs may not be optimal, and often are not feasible, for study of infections caused by drug-resistant bacteria. Over the past several years, multiple stakeholders have worked to develop consensus regarding paths forward with a goal of facilitating timely conduct of antimicrobial development. Here we advocate for a novel and pragmatic approach and, toward this end, present feasible trial designs for antibacterial agents that could enable conduct of narrow-spectrum, organism-specific clinical trials and ultimately approval of critically needed new antibacterial agents.
Posted by
Rima Mycynek

